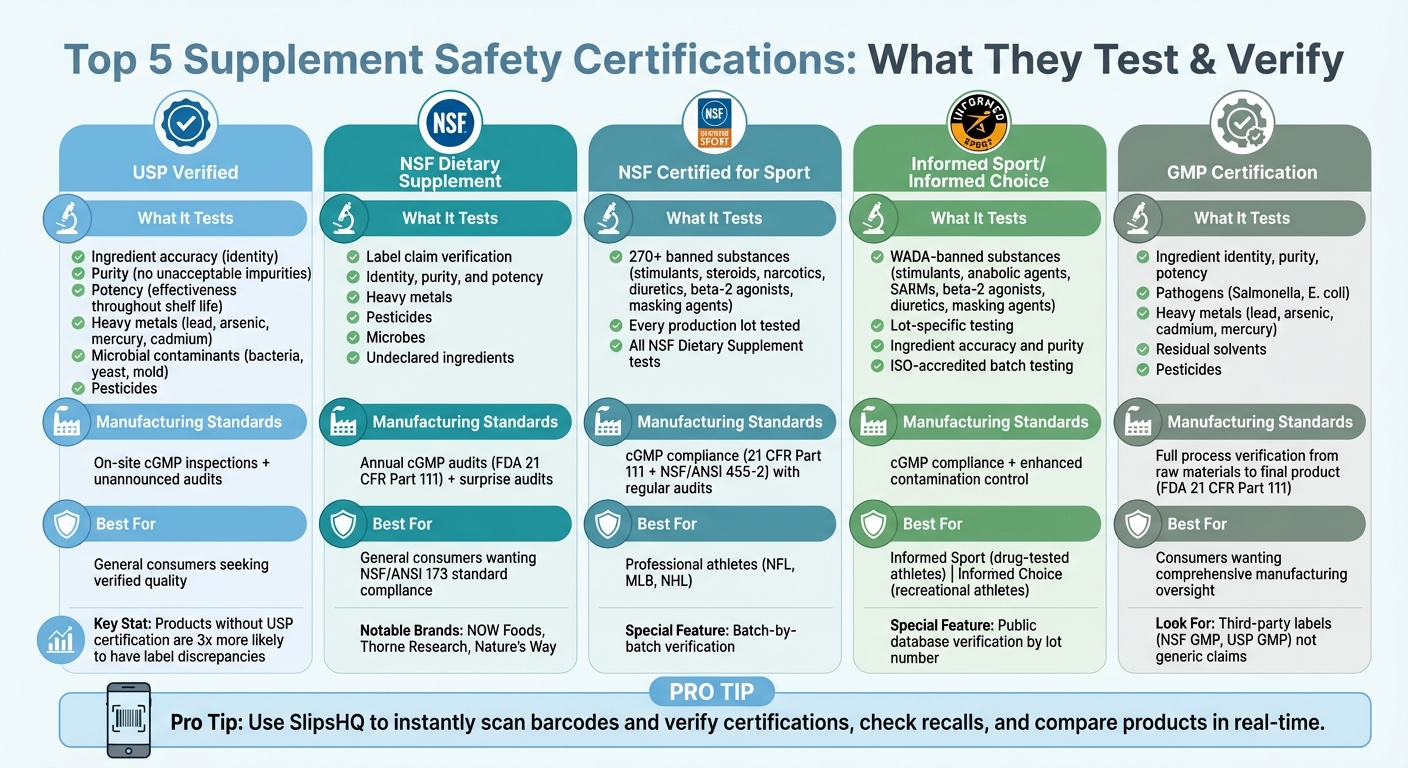
Top 5 Certifications for Supplement Safety

Looking for safer supplements? Certifications like USP Verified, NSF, and GMP can help you identify high-quality products. These seals confirm that a supplement matches its label, is free from harmful contaminants, and is produced in clean, regulated facilities. Here's a breakdown of the top certifications to know:
- USP Verified: Tests for ingredient accuracy, purity, and potency while ensuring compliance with manufacturing standards.
- NSF Dietary Supplement: Verifies label claims, screens for contaminants, and audits manufacturing facilities annually.
- NSF Certified for Sport: Adds banned substance testing for athletes, ensuring compliance with sports regulations.
- Informed Sport/Informed Choice: Focuses on drug-tested athletes by screening for prohibited substances and ensuring ingredient purity.
- GMP Certification: Covers manufacturing processes, from raw materials to final product, ensuring safety and consistency.
Pro Tip: Use tools like SlipsHQ to instantly verify certifications, check for recalls, and compare products. Pair certifications with these insights to make informed choices and prioritize your health.
Top 5 Supplement Safety Certifications Comparison Chart
1. USP Verified Mark
The USP Verified Mark, issued by the United States Pharmacopeia, stands as one of the most trusted symbols of quality in the supplement industry. This mark indicates that a product has undergone rigorous testing to confirm accurate ingredients, proper strength, and freedom from harmful contaminants.
Emphasis on Ingredient Accuracy, Purity, and Strength
USP certification zeroes in on three key aspects: identity (ensuring the ingredients match the label), purity (confirming the absence of unacceptable impurities), and potency (ensuring the product maintains its effectiveness throughout its shelf life). According to a 2023 study, supplements lacking USP certification were found to be up to three times more likely to have discrepancies between their labels and actual contents.
Screening for Harmful Contaminants
In addition to verifying ingredient integrity, USP certification includes thorough testing for potential hazards. This involves screening for heavy metals like lead, arsenic, mercury, and cadmium, as well as microbial contaminants such as bacteria, yeast, and mold. The process also checks for pesticides and other impurities, ensuring all levels fall below strict safety limits. These measures help guarantee that the product is not only effective but also safe for consumption.
Ensuring Compliance with Manufacturing Standards
USP doesn’t stop at testing the final product - it also evaluates the manufacturing process. Through on-site inspections, USP ensures facilities comply with current Good Manufacturing Practices (cGMP). These inspections cover quality control procedures, documentation, and overall facility cleanliness. Additionally, unannounced audits are conducted to confirm ongoing compliance, making USP certification a step above standard GMP claims.
2. NSF Dietary Supplement Certification
The NSF Dietary Supplement Certification operates under NSF/ANSI 173, the only American National Standard tailored specifically for dietary and nutritional supplement ingredients. This certification plays a key role in ensuring dietary supplements meet strict safety and quality standards, which is especially important in the U.S., where many products on the market lack independent testing.
Emphasis on Ingredient Identity, Purity, and Potency
NSF’s label claim review ensures that the ingredients listed on a product’s label are not only present but also in the correct amounts. Through detailed laboratory testing, NSF validates the identity, purity, and potency of the supplements. For instance, if a vitamin D supplement advertises 2,000 IU per capsule, NSF testing confirms that this dosage is accurate. Brands such as NOW Foods, Thorne Research, and Nature’s Way are among those that have earned NSF certification, with public listings available to verify their compliance.
Screening for Harmful Contaminants
NSF certification also involves a contaminant review, which screens for hazardous substances like heavy metals, pesticides, microbes, and undeclared ingredients. Certified products undergo periodic retesting to ensure they continue to meet safety standards.
Ensuring Compliance with Manufacturing Standards
To further enhance supplement safety, NSF conducts annual audits of manufacturing facilities to confirm compliance with current Good Manufacturing Practices (cGMP) as outlined in FDA 21 CFR Part 111. These assessments often include unscheduled, surprise audits to ensure facilities maintain proper quality control, accurate documentation, and cleanliness throughout the year. By evaluating both the products and the production processes, NSF certification goes beyond one-time checks, setting a higher standard for safety and reliability.
3. NSF Certified for Sport
This program builds on NSF's standard certification process but is tailored to meet the specific needs of athletes. While NSF's general certification ensures safety and accurate labeling, the NSF Certified for Sport program takes things further. It tests every production lot for over 270 banned substances that could lead to failed drug tests or disqualification.
Extra Safeguards for Athletes
The program screens for substances like stimulants, steroids, narcotics, diuretics, beta-2 agonists, and masking agents - compounds prohibited by major U.S. sports organizations, including the NFL, MLB, and NHL. Beyond banned substances, products are also checked for contaminants. For athletes competing under strict anti-doping regulations, this lot-by-lot testing minimizes the risk of accidental violations. Many professional teams and athletic programs rely on NSF Certified for Sport products to protect their athletes' careers and uphold their own reputations.
Ensuring Manufacturing Compliance
To keep the certification, products must be made in facilities that comply with cGMP under 21 CFR Part 111 and NSF/ANSI 455-2 standards. These facilities undergo regular on-site audits to review production records, quality control measures, raw material sourcing, and batch traceability. You can confirm certification by looking for the NSF mark on product labels or checking NSF’s public database using batch numbers. This certification serves as an additional layer of safety in supplement manufacturing and testing.
4. Informed Sport / Informed Choice Certification
Informed Sport and Informed Choice are certifications managed by LGC that focus on minimizing the risks associated with banned substances. Informed Sport is tailored for professional and competitive athletes who undergo drug testing, while Informed Choice caters to recreational athletes and fitness enthusiasts who want their supplements regularly screened. Both certifications are widely recognized by sports organizations in the U.S. and internationally as reliable anti-doping measures.
Extra Safeguards for Sports Supplements
One of the standout features of these certifications is their lot-specific testing for banned substances. Products undergo rigorous analysis to detect a wide range of substances prohibited by the World Anti-Doping Agency (WADA). This includes stimulants, anabolic agents, SARMs, beta-2 agonists, diuretics, and masking agents. Even trace amounts of these substances could result in a positive doping test, so this level of testing is critical. Athletes can confirm whether a specific product lot has been tested by referencing the Informed Sport database - an essential tool under strict anti-doping regulations.
Ensuring Ingredient Accuracy and Purity
Both certifications mandate thorough formulation reviews and ISO-accredited batch testing to confirm that ingredients are accurate, potent, and free from undeclared substances. This process ensures that athletes and consumers can trust the integrity of the products they use.
Meeting Manufacturing Standards
In addition to standard cGMP compliance, these certifications provide an added layer of contamination control. Brands are required to adhere to cGMP standards while also documenting their quality systems. Many sports supplement brands pair these certifications with others, like NSF or GMP credentials. While cGMP focuses on overall manufacturing quality and safety, the Informed programs specifically address the risks of banned substances for athletes.
For U.S. consumers, identifying certified products is simple: look for the Informed Sport or Informed Choice logo on the label. You can cross-check the product details - such as the brand name, product, flavor, and lot number - on the official certification website. Tools like SlipsHQ make this process even easier by allowing users to scan barcodes for instant access to third-party certifications, ingredient safety information, and warnings. This technology helps build a safer supplement routine with clear and accessible data.
5. Good Manufacturing Practices (GMP) Certification
Good Manufacturing Practices (GMP) certification plays a key role in ensuring supplement safety. Unlike certifications that focus solely on testing finished products, GMP addresses every step of the manufacturing process - from sourcing raw materials to bottling the final product. In the U.S., the FDA requires supplement manufacturers to comply with current Good Manufacturing Practices (cGMP) as outlined in 21 CFR Part 111, setting the legal minimum standard. However, third-party certifications from organizations like NSF and USP go beyond these requirements by independently verifying consistent adherence to these standards.
Verification of Compliance with Manufacturing Standards
Third-party GMP certification involves thorough audits of manufacturing facilities to confirm compliance with FDA cGMP regulations. These audits evaluate critical aspects such as employee training, equipment maintenance, detailed batch records, and recall protocols. Unlike vague claims like "made in a GMP facility", which may lack proper documentation, third-party certifications rely on regular renewals and unannounced inspections to ensure manufacturers remain compliant. This external validation provides a higher level of trust and accountability.
Emphasis on Ingredient Identity, Purity, and Potency
GMP standards require manufacturers to verify the identity, purity, and potency of their ingredients. This process involves supplier qualifications, certificates of analysis, and rigorous final-product testing. Third-party programs aligned with GMP, such as those from NSF and USP, audit these procedures to ensure consistency and reliability.
Testing for Harmful Contaminants
Another critical aspect of GMP certification is the proactive management of contaminants. Manufacturers are required to assess potential contamination risks and establish testing protocols for harmful substances. These tests often include checks for pathogens like Salmonella and E. coli, heavy metals such as lead, arsenic, cadmium, and mercury, as well as residual solvents and pesticides where applicable. Third-party certifiers review the facility's contamination control plans and may mandate routine batch testing to confirm that contaminant levels remain within safe limits. If a batch fails to meet these standards, it is quarantined to prevent distribution.
For U.S. consumers, finding GMP-certified products is relatively simple. Look for third-party labels like "NSF GMP" or "USP GMP" on product packaging or websites instead of relying on generic claims. Many certifiers offer public databases where you can verify certifications. Tools like SlipsHQ even allow you to scan a product's barcode to instantly access GMP certification details - making it easier to choose supplements with confidence and transparency.
How Digital Tools Work With Certifications
Independent seals like those from USP, NSF, and GMP programs confirm the quality of supplements, but keeping these certifications up to date and accessible can be a hassle. That’s where digital tools like SlipsHQ come in. Instead of relying on manual checks across various registries, SlipsHQ simplifies the process by converting static certification seals into real-time data. By scanning a supplement's barcode in the app, users can instantly verify the product’s certifications and audit dates through public and partner registries, including USP, NSF, Informed Sport/Choice, and accredited GMP programs. This seamless integration builds on the trust established by traditional certification systems.
The app goes a step further by incorporating certifications into a 0–100 trust score, which evaluates products based on 35 factors like safety, efficacy, quality, transparency, and value. Supplements with certifications from programs such as USP, NSF, or NSF Certified for Sport earn higher scores because these programs conduct label-claim verifications, contaminant screenings, and regular audits. Beyond certifications, SlipsHQ adds layers of data, including third-party lab reports, recall histories, and ingredient-level risk assessments. This allows the app to highlight distinctions, such as products with clear dosage transparency versus those with vague formulations, helping users make better-informed decisions.
For athletes, the app offers a tailored feature to filter products certified by NSF Certified for Sport or Informed Sport/Choice. Scanning a supplement that lacks proper sports certification triggers an immediate alert, flagging potential risks and suggesting certified alternatives. This real-time verification ensures athletes can build supplement stacks that comply with anti-doping regulations without the hassle of manually cross-checking each product.
Given the FDA’s post-market oversight, SlipsHQ combines certification data with real-time recall alerts and emerging safety updates to maintain an accurate product safety profile. Users can create personalized supplement stacks that prioritize certified options, compare prices across U.S. retailers in U.S. dollars, and get reminders based on specific label directions. The app also helps avoid redundant or potentially harmful ingredient combinations, making it a practical tool for health-conscious individuals.
With data on over 200,000 supplements and a stellar 4.9 rating, SlipsHQ enhances the value of traditional certifications by delivering instant, actionable insights. It doesn’t replace programs like USP or NSF but complements them, empowering U.S. consumers to make smarter, data-driven choices. After a 3-day free trial, the app costs $4.99 per week or $39.99 per year - a small price for peace of mind and smarter supplement decisions.
Conclusion
Certifications like USP Verified, NSF Dietary Supplement, NSF Certified for Sport, Informed Sport/Informed Choice, and GMP act as your first safeguard when selecting supplements. These seals confirm that the product matches its label claims, has been tested for contaminants like heavy metals and microbes, and is produced in facilities adhering to stringent quality controls. This level of assurance goes beyond the basic FDA oversight, which does not pre-approve supplements before they hit the market.
While these certifications provide a reliable foundation, digital tools can simplify and enhance the process of verifying supplements. Certifications focus on safety and manufacturing standards but don’t guarantee the product fits your specific health needs. That’s where tools like SlipsHQ come in. By scanning a product’s barcode, the app instantly reveals its certifications, flags any ingredient safety concerns, identifies doses outside evidence-based ranges, and even compares prices across U.S. retailers - all in one convenient platform.
When buying supplements, start by checking for at least one reputable certification. Next, use SlipsHQ to analyze your current supplements, uncover potential risks, and streamline your routine. Finally, discuss your choices and SlipsHQ’s findings with your healthcare professional to ensure they align with your health goals and budget. This three-step process - 1. Verify certifications, 2. Use SlipsHQ for detailed product insights, 3. Consult your healthcare provider - offers a practical way to make informed decisions in the often-overwhelming supplement market. With trusted certifications and digital insights working together, you can confidently navigate your options and prioritize your well-being.
FAQs
How is USP Verified different from other supplement certifications?
USP Verified sets itself apart with a strict, science-backed evaluation process to confirm that supplements meet high standards for safety, quality, and purity. This ensures that products include the ingredients listed on their labels at the stated potency while being free from harmful contaminants.
This certification empowers consumers to make informed decisions by providing clear information about ingredient safety, potential interactions, and overall product integrity. It’s a trusted mark for those who value supplement safety and reliability.
What does NSF Certified for Sport mean, and how does it ensure supplement safety for athletes?
The NSF Certified for Sport label is a trusted mark of safety and quality in the world of supplements. It ensures that products meet stringent standards designed specifically for athletes. Supplements with this certification undergo thorough testing to guarantee they are free from banned substances, harmful contaminants, and unsafe ingredients.
For athletes, this certification provides peace of mind. It confirms that the supplements they’re using are not only safe but also compliant with sports regulations. Plus, it ensures these products are manufactured under strict quality controls. In a supplement market that can often feel overwhelming, this certification helps athletes make smarter, safer choices.
What makes GMP certification essential for supplement safety?
GMP, or Good Manufacturing Practices, certification plays a key role in ensuring that supplements are made under strict quality standards. This certification helps minimize the risk of contamination, ensures ingredient labels are accurate, and prioritizes consumer safety.
When manufacturers follow GMP guidelines, they show a clear dedication to creating reliable, high-quality products that comply with regulatory standards. For consumers, this certification offers confidence in choosing supplements that support their health and well-being.
